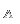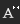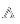Crystal Structure – Atomic Arrangements and Crystal Lattices
Minerals can be formed directly from lava or volcanic action. They can also be formed through weathering, evaporation as well as the settling of hydrothermal solutions. Changes in temperature and pressure can trigger alteration as well. Despite the differences in mineral formation environments, every mineral not only has its own particular range of chemical composition it is also arranged in a specific geometric way inside. We call this the "Crystal Structure".
Naturally occurring minerals may vary in their atomic or ionic structure, or have different crystal structures, but where the geological conditions are right the homogenous solids formed by these atoms or ions all have regular and symmetrical crystal structures. These along with the smooth facets that form on the surface allow all kinds of different "Mineral Crystals" to form.
People began analyzing the internal structure of mineral crystals in the early 20th Century using X-rays. Only then were they able to work out the true arrangement of the crystal structure and particles (atoms, ions or molecules). The studies revealed that regardless of what the mineral or crystal looked like externally, the internal particles all followed a set arrangement that in turn determines the shape unique to that crystal.
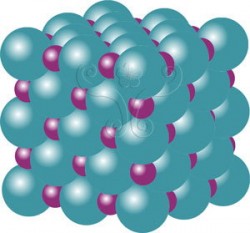
Native copper for example is made up of copper atoms stacked tightly in a cubic manner. The atoms are located at the corners and the center of each face of the cubic cell. Another example is rock salt. The chemical composition is sodium chloride and in Image 2, the large green ball and the small red ball represent the Chlorine (Cl-) and Sodium (Na+) irons. These particles always appear in a three-dimensional space at set intervals. If we use a solid circle and a hollow circle to represent the center of the chlorine and sodium ions then link them together with straight lines, we end up with a cubic lattice. This is the reason why rock salt crystals usually appear as cubes.
Depending on the composition of each mineral and the way their atoms or ions are stacked, the result is a complex world of minerals filled with many different varieties and compositions.
Crystal System – the Highest Common Factor of the Mineral World
The atoms within crystalline minerals always follow a set arrangement and this in turn determines the unique shape of the crystal. The geometric characteristics of a mineral crystal's atomic structure or appearance are usually described using the coordinate system. In the coordinate system the three vectors of the crystal are defined using the letters a, b and c. These are referred to as the crystallographic axes. The angle between the three axes are indicated by α, β and γ. Crystal constants such as axes length and the included angles allow crystal minerals to be divided into six basic types known as the "Crystal System".
The geometric characteristics of each crystal system and their representative mineral are described below:
1. Isometric System – Also known as the Cubic System. All three axes are of equal length and perpendicular to each other. Example: Diamond, Garnet.
2. Tetragonal System – Has three axes perpendicular to each other. Two of the axes are of equal length, with the third perpendicular axes being of a different length. Example: Zircon, Rutile.
3. Hexagonal System – Has four crystal axes with three parallel axes of equal length that intersect with each other at an angle of 60°. Another axis intersects the parallel axes perpendicularly and is of a different length. Example: Beryl, Quartz.
4. Orthorhombic System – Has three crystal axes all of different lengths that are perpendicular to each other. Example: Sulfur, Olivine.
5. Monoclinic System – Has three crystal axes a, b and c all of different lengths. Axis b is perpendicular to the other two axes while axes a and c intersect each other at an angle not equal to 90°. Example: Gypsum, Diopside.
6. Triclinic System – Has three crystal axes all of different lengths that all intersect each other at an angle. Example: Kyanite, Albite.
Crystal Plane Symbol – Spatial Notations for Crystal Planes
In books on mineralogy or crystal formation, you will often see three to four Arabic numerals in parentheses noted above each plane of a crystal. The symbol is used to indicate the spatial relationship between a mineral crystal's crystal planes so it's known as the "Crystal Plane Symbol". The crystallographic axis is used as the reference axis (imaginary line) together with the axial units above to indicate the location of the crystal plane. This is made up of 3 to 4 (cubic and hexagonal) coprime small integers in parentheses. As the notation was created by the British mineralogist Miller in 1839, it is also known as the "Miller Index".
The Miller Index for any crystal is equivalent to the inverse intercepts of that crystal plane on three or four crystallographic axes. In Image 1 for example, to find out the symbol for crystal plane ABC you must first calculate the inverse intercepts for plane ABC along the X, Y and Z axes. The result is:
1/2:1/3:1/6=3:2:1 (The three smallest coprime integers)
The symbol for plane ABC is therefore (321).
In the standard coordinate system, the front of the X-axis is positive and behind is negative; the right side of the Y-axis is positive and the left is negative; in the Z-axis, up is positive and down is negative. If a crystal plane intersects with the negative end of a crystallographic axis, a bar must be marked above the corresponding index, so in Image 1 plane ACD's symbol is (321).
In the Crystal System the hexagonal system is unusual in that its Miller Index is made up of 4 smallest coprime integers. As a result, Beryl's Miller Index is as shown in Image 2.
Using the Isometric System as an example, the crystal in Image 3 is made up of cubic and octahedral lattices. Can you find the correct symbol for each crystal plane?
National Museum of Natural Science
|